COVID-19 Effect on Pharmaceutical Innovation

Sarbani Chattopadhyah
March 28, 2022
Check out the 2023 version of the Innovation Momentum report, available now!
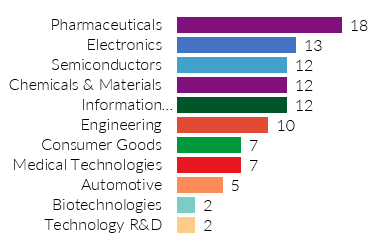
As the world reaches the 2-year milestone of the COVID-19 pandemic, we have witnessed remarkable developments in efforts to fight the virus. Whether in the form of preventative measures or medical treatments, researchers and scientists have had excellent innovation momentum in delivering solutions for one of the most significant crises of our time. Big Players in these advancements are pharmaceutical companies. We see the pharmaceutical innovation reflected in a new report, “Innovation Momentum 2022: The Global Top 100.“ Pharmaceuticals is the most represented industry on the list, with 17 companies in the top 100.
The following is a deep dive into the pharma innovators and details about the specific technologies these companies have been developing.
The innovation momentum methodology and Patent Asset Index
The Innovation Momentum 2022: The Global Top 100 report uncovers forward-looking technological developments and spotlights true innovative contemporaries across all industries. The Innovation Momentum methodology follows the Patent Asset Index, an industry-trusted, scientifically developed index, a part of LexisNexis® PatentSight®, created to assess patents based not only on size but also the individual quality of all patent families belonging to a patent portfolio.
The Innovation Momentum approach is unique because it:
- Recognizes patent owners with relatively small but high-quality portfolios
- Rewards patent owners with well-maintained portfolios
- Reflects the dynamics of thriving technologies
- Considers the differences of developments in all technology fields
- Considers the innovators’ target markets.
The 17 Pharmaceutical companies with the strongest innovation momentum
Pharmaceuticals is the largest industry sector with a total of 17 innovators in the Top 100 and strongly highlights a theme of focus across all industries in this year’s Top 100: the COVID-19 pandemic.
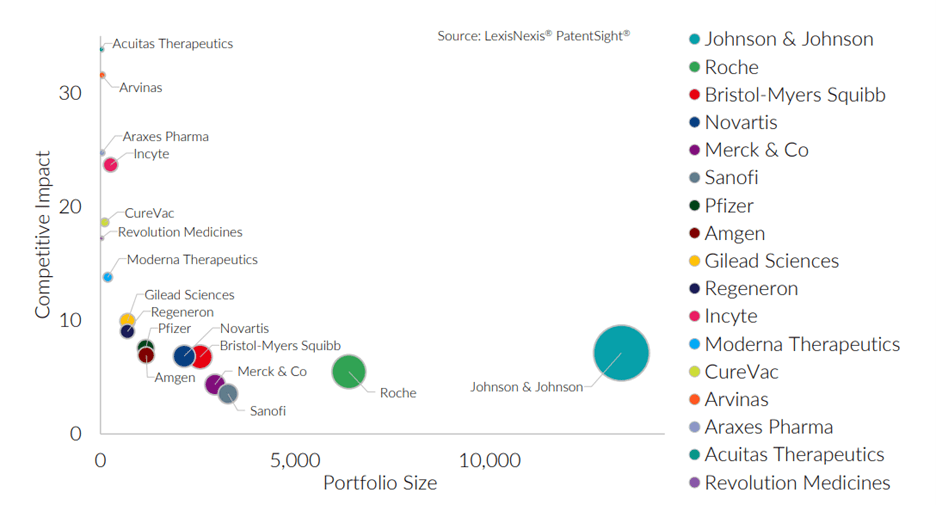
Johnson & Johnson and Roche both have the most robust portfolios in this sub-technology. These two have the largest portfolios in the technology group and, like the others, have a Competitive Impact above the database average of about one. They have increased their patent portfolio strength over recent years, further improving the quality of their patents.
This chart shows the average patent quality (Competitive Impact) versus Portfolio Size for pharmaceuticals industry sector patent owners. The bubble size for the patent owners scales with portfolio strength (Patent Asset Index).
Pharmaceutical players and their COVID-19 highlights
Even before COVID-19 reached official pandemic levels in 2020, pharmaceutical companies were working on technologies that provide both preventative mechanisms and better treatment options for coronavirus illness. This innovation enabled the health community to quickly mobilize resources when the virus reached critical global infection rates. The following are some of the contributions of the innovators on the 2022 list.
Johnson & Johnson has the strongest patent portfolio in the Innovation Momentum Report pharmaceutical sector.
In February 2021, the single-dose Janssen COVID-19 vaccine candidate from Johnson & Johnson was authorized for Emergency Use Authorization (EUA) by the U.S. Food and Drug Administration.
This COVID-19 vaccine leverages the company’s AdVac® technology platform which was also used to develop and manufacture the Ebola vaccine and construct its Zika, RSV, and HIV vaccine candidates.
Roche developed the first commercially distributed diagnostic test to receive an EUA during the COVID-19 outbreak. The company said that “developing and getting regulatory approval for a new diagnostic usually takes 12 to 18 months”, but Roche was able to do this in about six weeks.
Gilead Sciences‘ broad-spectrum antiviral drug Remdesivir has been used as an emergency treatment of COVID-19 infected patients since the pandemic outbreak.
Sanofi, in collaboration with GlaxoSmithKline (GSK), also has a COVID-19 vaccine in the pipeline that comprises the spike protein with the adjuvants that are the respective proprietary property of Sanofi and GSK.
Moderna has contributed to the development of modified messenger RNA (mRNA) medicines and is a leader in the space. The years of research and innovation around mRNA allowed Moderna to finalize the sequence for mRNA-1273 within days of receiving the genetic sequence of the virus.
CureVac has also seen some recognition working on mRNA-based therapies. According to a recent news statement, their multivalent technology approach for seasonal influenza mRNA vaccine candidate addresses four different influenza strains.
Pfizer is a notable appearance on the 2022 list—at the end of 2021, the U.S. Center for Disease Control (CDC) recommended the Pfizer-BioTech pediatric COVID-19 for 5–11-year-olds.
Acuitas Therapeutics is a Canadian company whose lipid nanoparticle delivery system is used by Pfizer for their COVID-19 vaccine. The two entities have entered into an agreement under which Pfizer will have the option to license, on a non-exclusive basis, Acuitas’ LNP technology for up to 10 targets for vaccine or therapeutic development.
Strong innovation momentum of players focused on cancer treatment
While COVID-19 has been the most visible medical crises of the past 2 years, there are still a wide variety of health challenges that are being studied, with innovative outcomes. This is highlighted with the progress made by the other pharmaceutical groups on the Innovation Momentum list.
Amgen has been active in the field of cancer immunotherapy for some time now and has invented path breaking potential treatments against different types of cancers, the most recent being lung cancer.
Araxes Pharma has been developing cancer drugs in collaboration with Janssen for some time now and has also cleared an investigational new drug (IND) application targeting non-small lung cancer.
Arvinas is the newly emerging player in cancer immunotherapy concentrating on Interleukin therapy.
Bristol-Myers Squibb has gotten the approval from the U.S. Food and Drug Administration for a groundbreaking immunotherapy-based treatment for non-small cell lung cancer.
Incyte is another company making strides in the field of cancer immunotherapy through its proprietary inventions.
Merck holds a strong position in cancer therapy with its patented cancer drugs and has been able to utilize its know-how in antivirals to bring oral antiviral medicine for non-hospitalized adult patients with mild-to-moderate COVID-19.
Novartis has focused on treating diseases like psoriasis, muscular atrophy, and cancer. It has also ventured into precision treatment, as indicated by its patent portfolio.
Regeneron is another front runner in the attempt to bring mRNA-based vaccines and drugs to the market.
Revolution Medicines has been targeting cancers caused by specific genetic mutations.
Pharmaceutical innovation in 2022 and beyond
Getting a brief look at the impressive technological innovation represented in the pharmaceuticals category of “Innovation Momentum 2022” provides more insight into the value of the methodology and measuring patent quality over quantity. The list and its approach give us a chance to recognize these innovators’ significant contributions, many of whom are improving lives with their research and development.
See the top 100 innovators list by reading the full report, Innovation Momentum 2022.
Learn more about PatentSight®.

Get actionable insights for strategic decision making
Want to become more effective at managing your patent portfolio, identify worldwide relevant patents and technology trends, assess the competitive landscape, and find partners and licensing opportunities?
